Abstract
Introduction:
Underdiagnosis of COPD appears to be common, although the degree of underdiagnosis is rarely measured. To document the extent of underdiagnosis in a high risk group of ambulatory patients, we performed spirometry in smokers aged 40 years and over drawn from general practices in two countries.
Methods:
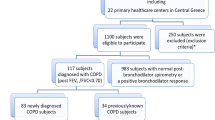
Subjects were recruited from primary care practices in Aberdeen, Scotland, and Denver, Colorado, via random mailing. Current and former smokers aged 40 or older with no prior diagnosis of chronic obstructive respiratory disease (and no respiratory medications within the past year) were enrolled. Participants underwent pre- and post-bronchodilator spirometry. A study diagnosis of COPD was defined as post-bronchodilator FEV1/FVC < 0.70.
Results:
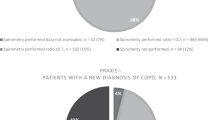
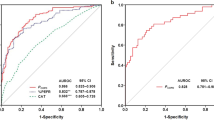
Spirometric examination was complete in 818 patients, of whom 155 (18.9%) had a study diagnosis of COPD. Using the Global Initiative for Chronic Obstructive Lung Disease (GOLD) severity criteria, the COPD was mild in 57.4%, moderate in 36.8%, and severe in 5.8%. No patients had very severe disease according to GOLD criteria.
Discussion:
Screening of smokers over 40 in general practice may yield 10 — 20% undiagnosed COPD cases, with a substantial proportion of these having moderate to severe disease. Earlier diagnosis through targeted case-finding will allow early, aggressive smoking cessation efforts and may lead to a reduction in the burden of COPD symptoms and a reduced impact of the disease on health-related quality of life in these patients.
Similar content being viewed by others
Article PDF
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
DGT is an employee of the National Jewish Medical and Research Center which received funding from Boehringer Ingelheim to participate in this study.
DBP has received honoraria for speaking at sponsored meetings and serving on advisory panels for the following companies marketing COPD products: AstraZeneca, Boehringer Ingelheim, GlaxoSmithKline, Novartis, and Pfizer. He or his research team have received funding for research projects from the following companies marketing COPD products: AstraZeneca, Boehringer Ingelheim, GlaxoSmithKline, Novartis, and Pfizer.
At the time this study was performed, RJN and RJH were employees of Protocare Sciences which provided consulting services to the pharmaceutical industry, including the sponsors of this study.
Rights and permissions
About this article
Cite this article
Tinkelman, D., Price, D., Nordyke, R. et al. COPD screening efforts in primary care: what is the yield?. Prim Care Respir J 16, 41–48 (2007). https://doi.org/10.3132/pcrj.2007.00009
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.3132/pcrj.2007.00009
This article is cited by
-
Accuracy of the COPD diagnostic questionnaire as a screening tool in primary care
BMC Primary Care (2022)
-
Identifying airway obstruction in primary care: is there a role for physiotherapists?
BMC Primary Care (2022)
-
A scoring system to detect fixed airflow limitation in smokers from simple easy-to-use parameters
Scientific Reports (2018)
-
GP utilisation by education level among adults with COPD or asthma: a cross-sectional register-based study
npj Primary Care Respiratory Medicine (2016)
-
Identifying cases of undiagnosed, clinically significant COPD in primary care: qualitative insight from patients in the target population
npj Primary Care Respiratory Medicine (2015)